
Richard Spurr 1am - 4am
14 March 2023, 12:04 | Updated: 14 March 2023, 17:15

Cold and flu remedies Day & Night Nurse capsules and Covonia are among 20 remedies pulled from UK shelves over "very rare" reactions, first flagged in Europe months ago.
The remedies - as well as own-brand version sold by pharmacies including Boots and Superdrug - have now been withdrawn from shelves "as a precaution" following a review by medical regulators.
The European Medicines Agency recommended their withdrawal three months ago, with the products' removal only taking place today.
Pholcodine is an opioid medicine found in a range of household cough syrups, with the immediate recall encompassing both syrups and lozenges across the UK.
The opioid is widely used in cold and flu products due to its ability to suppress coughing.
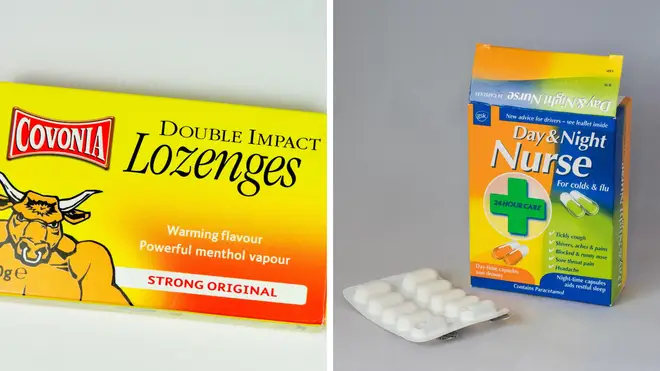
Regulators have now warned people to check the packaging of cough tablets and syrups at home to see if pholcodine is listed.
The review revealed the benefits of a drug named pholcodine, contained within the cough and cold medicines, "do not outweigh the increased risk of the very rare event of anaphylaxis".
The Medicines and Healthcare products Regulatory Agency (MHRA) polices the drug safety in Britain, however, the recall following a review has come three months after warnings were first raised across Europe.
Read more: Watch shocking moment woman smashes through wine bottles with frying pan in Tesco
Read more: Primary school teacher stabbed 'cheating' boyfriend to death and buried body in garden, court told
The MHRA flagged the evidence suggested an increased risk of anaphylaxis extended to "patients who receive general anaesthesia involving neuromuscular blocking agents during surgery".
PAGB Chief Executive, Michelle Riddalls, said: “We and our members put an absolute priority on patient safety. The risk in people who have used pholcodine is extremely small and the MHRA has taken this decision as a precaution.
“This precautionary measure is an example of the robust safety measures that we have in place in the UK working effectively.
“Our members are working very closely with MHRA, healthcare professionals and pharmacists to ensure that appropriate action is taken.
“Pholcodine containing products have only been available for purchase in pharmacies and if people have any questions pharmacists are well placed to give advice and recommend alternative products.”

Products containing Pholocodine are currently only available to purchase from UK pharmacies, as opposed to supermarket aisles.
The watchdog also pointed out there was "no increased risk" of allergic reactions with allergens other than specific neuromuscular blocking agents following the use of pholcodine.
"The absolute risk in patients who have used pholcodine is very small, but patients should talk to a pharmacist, their GP or their surgical team if they have any questions," the MHRA said.
“The Commission on Human Medicines (CHM), the independent advisory body that provides expert advice on the safety, quality and efficacy of medicines, has considered the evidence of an increased risk of the very rare event of anaphylaxis when exposed to neuromuscular blocking agents (NMBA) and advised that pholcodine-containing medicines should be withdrawn.”
The full list of products slated for recall are as follows:
Source: MHRA